B7-H3 is expressed in certain tumor cells in a variety of cancers, but undetectable or low in normal tissue
- B7-H3 expression is undetectable or low in normal tissue1–4
- B7-H3 expression may be associated with a poor prognosis for a variety of tumor types4–13

Diagram is for illustrative purpose only and does not represent a specific tumor type.
Was this informative?
B7-H3 expression is prevalent in certain human cancers
- B7-H3 is a transmembrane protein that belongs to the B7 family that includes immune checkpoint molecules such as PD-L114-17
- B7-H3 plays a role in antitumor immunity and regulating T cell activation17–20
- The receptor for B7-H3 is currently unknown, but is thought to be expressed by T cells17,21
-
B7-H3 was expressed in 72% (n=1,342) of patients with certain cancers34,a
- aIHC analysis using an anti-B7-H3 primary antibody (R&D Systems AF1027) of tumor cells from 1,342 commercially available human tumor tissues derived from a variety of cancer types. 72% of samples had detectable B7-H3 expression (IHC 1+, 16%; IHC 2+, 26%; IHC 3+, 30%)34
Click the buttons in the human body figure below to review certain cancers in which B7-H3 is expressed
The data shown in this section are from various publications derived from a review of the literature. Studies were only included if they used immunohistochemistry to measure B7-H3 protein expression. The details of each study can be found in the footnotes for each tumor type. The information shown comprises selected data and does not account for all evidence that may be available; therefore, no definitive conclusions should be drawn from these data.

Central nervous system
Head and neck
Thyroid
Esophageal
Lung
Breast
Melanoma
Gastric
Liver
Biliary tract
Pancreatic
Kidney
Colorectal
Ovarian
Prostate
Bladder
Cervical
Endometrial
Orthopedic
Orthopedic
Was this informative?
B7-H3 may exert pro-tumorigenic effects
- The function of B7-H3 remains under investigation; however, some data support that B7-H3 may exert pro-tumorigenic effects17
- Although the receptor for B7-H3 is unknown, there are several signaling pathways mediated by B7-H3 that have been identified17
- Preclinical models suggest that B7-H3 may initiate signaling through investigated cancer-associated pathways, such as PI3K/AKT/mTOR, Jak/STAT3, MVP/Raf/MEK, and NF-kB/VEGF17
- Through these signaling pathways, B7-H3 may exert pro-tumorigenic effects including promotion of cell proliferation, migration and invasion, immune evasion, and angiogenesis17
Click the buttons in the figure below to learn more about each investigational pathway.
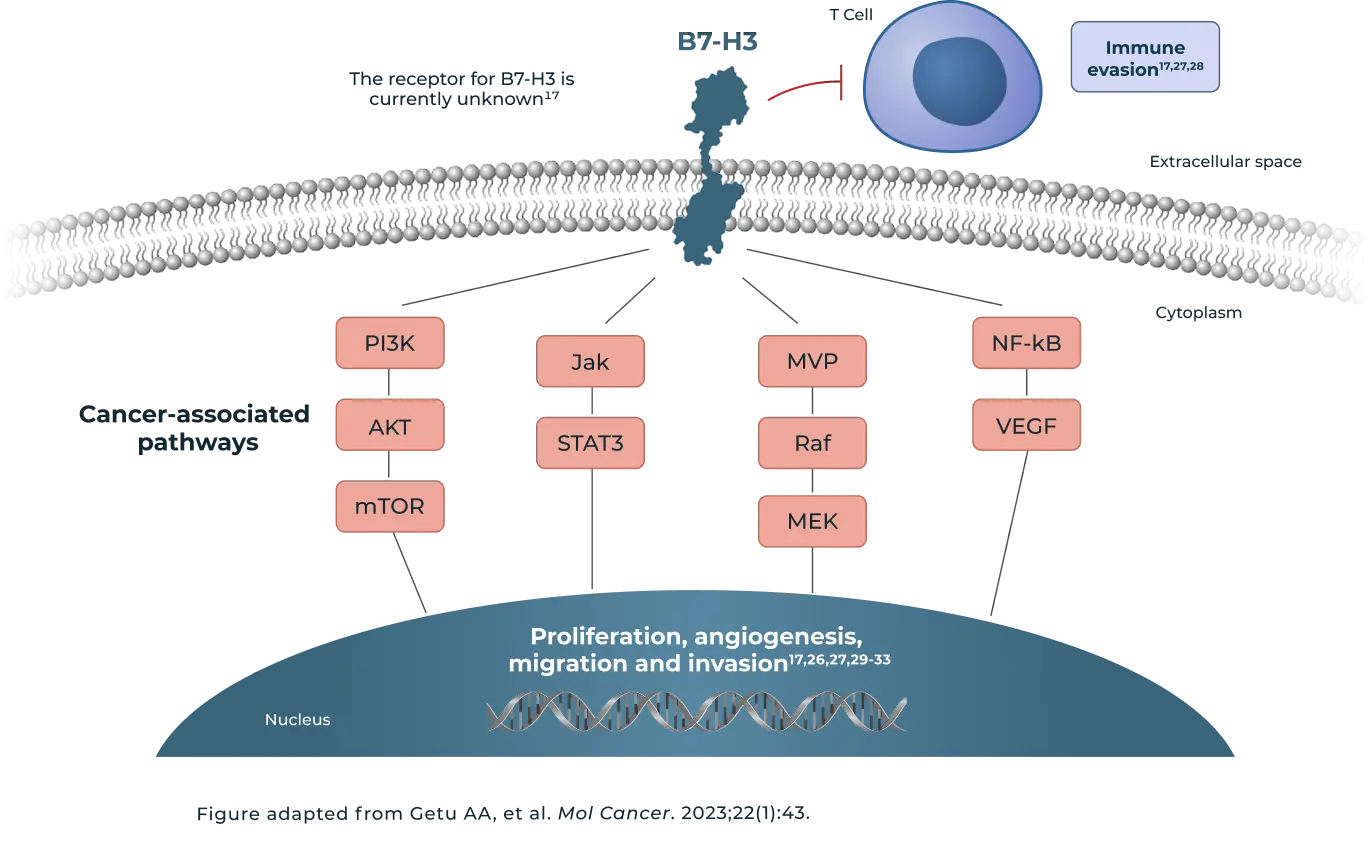
Upregulation of B7-H3 may activate PI3K and Jak/STAT3 signaling pathways to promote tumor cell migration and invasion, sustained tumor cell proliferation, and treatment resistance17,22–24
Upregulation of B7-H3 may activate PI3K and Jak/STAT3 signaling pathways to promote tumor cell migration and invasion, sustained tumor cell proliferation, and treatment resistance17,22–24
B7-H3 has been shown to initiate signaling through Raf/MEK through associations with major vault protein (MVP) and induce cancer stem cell and tumor formation25
Signaling through NF-kB activates VEGF to stimulate angiogenesis, which promotes cell survival and proliferation17,26
Cell proliferation – B7-H3 has been reported to activate signaling pathways such as ERK, PI3K, and STAT3 in certain cancer cells, which may lead to accelerated cell proliferation and tumor growth17
Angiogenesis – B7-H3 is shown to be implicated in angiogenesis in certain cancers. B7-H3 promotes angiogenesis by stimulating the secretion of VEGF17,26,29,30
Invasion and migration (metastasis) – B7-H3 function in tumor biology is linked to its ability to induce increased metastasis-promoting cell migration and invasion27,31-33
Immune evasion – B7-H3
expression is enriched within certain tumor microenvironments, where it facilitates immune evasion through various mechanisms and promotes an immunosuppressive microenvironment17,27,28
Was this informative?
Abbreviations
Akt, protein kinase B; B7-H3, B7 homolog 3; CNS, central nervous system; CRC, colorectal cancer; ER, estrogen receptor; ERK, extracellular signal-regulated kinase; ESCC, esophageal squamous cell carcinoma; HCC, hepatocellular carcinoma; HER2, human epidermal growth factor receptor; HNSCC, head and neck squamous cell carcinoma; ICC, intrahepatic cholangiocarcinoma; IHC, immunohistochemistry; Jak, Janus kinase; MEK, mitogen-activated protein kinase kinase; mNSCLC, metastatic non-small cell lung cancer; mTOR, mammalian target of rapamycin; MVP, major vault protein; NF-kB, nuclear factor kappa-light-chain-enhancer of activated B cells; NSCLC, non-small cell lung cancer; PDAC, pancreatic ductal adenocarcinoma; PD-L1, programmed cell death ligand 1; PI3K, phosphoinositide 3-kinase; Raf, rapidly accelerating fibrosarcoma protein; RCC, renal cell carcinoma; STAT3, signal transducer and activator of transcription 3; TME, tumor microenvironment; TNBC, triple negative breast cancer; VEGF, vascular endothelial growth factor.
References
1. Yang S, et al. Int J Biol Sci. 2020;16(11):1767-1773. 2. Dong P, et al. Front Oncol. 2018;8:264. 3. Yamato M, et al. Mol Cancer Ther. 2022;21:635-46. 4. Wang L, et al. Tumour Biol. 2016;37(3):2961-71. 5. Qiu M, et al. Front Oncol. 2021;11:600238. 6. Brunner A, et al. Gynecol Oncol. 2012;124(1):105-11. 7. Chen S, et al. J Cancer Res Clin Oncol. 2023;149(18):16609-16621. 8. Cong F, et al. Oncol Lett. 2017;14(6):7185-7190. 9. Song J, et al. Onco Targets Ther. 2016:9:6257-6263. 10. Katayama A, et al. Int J Oncol. 2011;38:1219-1226. 11. Wang L, et al. PLoS One. 2013;8(8):e70689. 12. Amori G. Prostate Cancer Prostatic Dis. 2021;24(3):767-774. 13. Inamura K, et al. Lung Cancer. 2017;103:44-51. 14. Zhong C, et al. Clin Exp Immunol. 2020;200(1):12-21. 15. Khan M, et al. Front Immunol. 2021;12:651634. 16. Pardoll DM. Nat Rev Cancer. 2012;12(4):252-64. 17. Getu AA, et al. Mol Cancer. 2023;22(1):43. 18. Kobori H, et al. Immunology. 2010;130(3):363-373. 19. Luo L, et al. J Immunol. 2004;173:5445-50. 20. Leitner J, et al. Eur J Immunol. 2009;39:1754-1764. 21. Chapoval AI, et al. Nat Immunol. 2001;2(3):269-274. 22. Wu X, et al. Transl Cancer Res. 2024;13(2):833-846. 23. Sun M, et al. J Cancer. 2021;12(24):7518-7526. 24. Zhou L and Zhao Y. Cancer Manag Res. 2019:11:10205. 25. Liu Z, et al. Oncogene. 2019;38(1):88-102. 26. Wang R, et al. Cell Death Dis. 2020;11(1):55. 27. Kanchan RK, et al. Biochim Biophys Acta Rev Cancer. 2022;1877(5):188783. 28. Gay CM, et al. Clin Cancer Res. 2025;31(16):3476-3482. 29. Cheng R, et al. APMIS. 2018;126(5):396-402. 30. Cheng N, et al. Biochem Pharmacol. 2021:183:114298. 31. Chen YW, et al. Curr Cancer Drug Targets. 2008;8(5):404-41. 32. Li Y, et al. Oncotarget. 2017;8(42):71725-71735. 33. Liu F, et al. Mol Med Rep. 2015;12(4):5455-5460. 34. Seaman S, et al. Cancer Cell. 2017;31(4):501-515. 35. Nehama D, et al. eBioMedicine. 2019;47:33-43. 36. Borgmann M, et al. J Cancer Res Clin Oncol. 2023;149(7):3383-3393. 37. Yonesaka K, et al. Clin Cancer Res. 2018;24(11):2653-2664. 38. Boland JM, et al. Clin Lung Cancer. 2013;14(2):157-63. 39. Joshi V, et al. Int J Mol Sci. 2024;25(7):3976. 40. Guo L, et al. Medicine (Baltimore). 2019;98(8):e14663. 41. Sun TW, et al. Cancer Immunol Immunother. 2012;61(11):2171-82. 42. Liu CL, et al. Eur Rev Med Pharmacol Sci. 2016;20(21):4466-4473. 43. Chen X, et al. Arch Pathol Lab Med. 2023;147(4):465-473. 44. Lu Z, et al. Mod Pathol. 2020;33(11):2330-2340. 45. Özalp FR, et al. BMC Cancer. 2024;24(1):1471. 46. Boorjian SA, et al. Clin Cancer Res. 2008;14(15):4800-8. 47. Zong L, et al. Mod Pathol. 2022;35(6):786-793. 48. Bonk S, et al. Pathol Int. 2020;70(10):733-742. 49. Lynch MM, et al. BMC Cancer. 2024;24:1336. 50. Zhang Q, et al. BMC Cancer. 2022;22:1124. 51. Zhao B, et al. Front Cell Dev Biol. 2022;10:819236.
Click to review reference list




